Get Tech Tips
Subscribe to free tech tips.
Adiabatic Cooling, Blower Settings, and Why You Care
Just so you don't get bored and quit reading, let's get straight to the point.
When the blower runs for more than a few minutes after the system has cycled off in cool mode, the air may continue to be “cooler” (lower sensible temperature) coming out of the supply. However, the heat content of the air will remain unchanged.
The only reason I say “may” be cooler instead of “will” is because we're assuming there is moisture on the coil or in the pan, and the indoor RH is less than 100%.
Translation: When you run the blower once the system has gone off in cool mode, you will continue to cool for a while, but that extended cooling comes from the evaporation of water off of the coil and out of the pan. That results in sensible cooling and greater sensible efficiency. However, it will also result in increased indoor humidity.
Translation of the translation: It may feel cooler, but there isn't any less heat in the air by the time you figure for humidity. (Remember, there are latent BTUs, not just sensible.)
Translation of the translation's translation: If you live in a humid place, run shorter off-cycle run times; you'll also want to think twice before running the fan in the “on” position. If you are in a dry place, then let it blow to your heart's content.
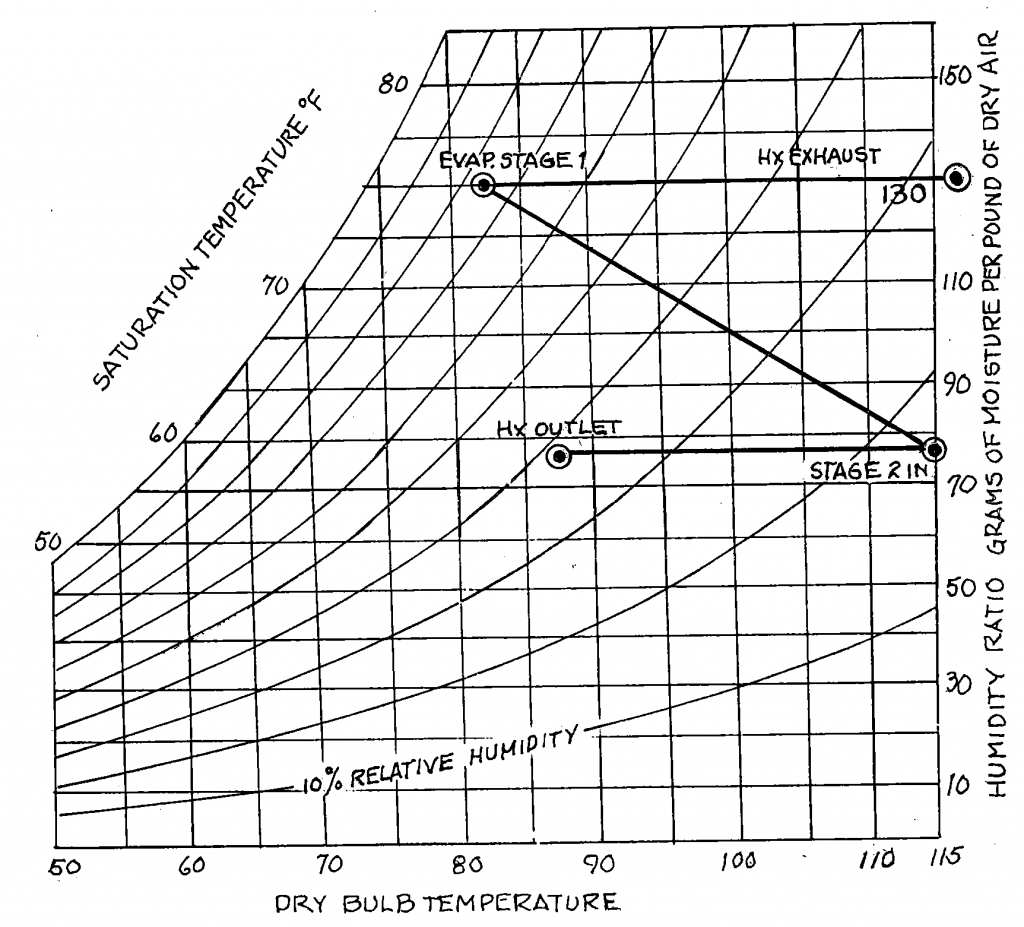
Whenever cooling occurs by direct evaporation of a substance into an airstream (think a swamp cooler), it occurs at no net decrease to the heat content in the air. The heat is just going from sensible (what you can measure with a thermometer) to latent. As a result, you get air with higher relative humidity.
If you go below this line, it is going to get nerdy—BEWARE!
The Nerdy Side
Now, let's talk about why all this happens. But first, let's cover some terms.
Heat = Molecular energy or total molecular movement within a substance
Temperature = Molecular velocity, the speed that the molecules are moving
Adiabatic Process = A change in temperature without a change in heat content
Think of an adiabatic process like this:
You have a whole room full of ping pong balls bouncing around in a zero-gravity room. The balls are molecules; their total motion is the amount of heat, and their speed is temperature. If you were to change the size of the room by bringing in one of the walls, the balls would bounce faster because the available space was decreased. Therefore, the “temperature” would increase. However, the number of balls and the total motion would remain constant. (This example also represents what happens to the refrigerant in a compressor, by the way.) If you were to move a wall outward and increase the size of the room, the speed of the molecules would decrease; you'd get less speed and lower “temperature.” All the while, the number of balls and the total motion remained constant. (This example occurs at the outlet of the metering device).
In both examples, temperature (sensible heat) changes, but the total heat content does not change; these are examples of an adiabatic process due to compression and expansion of contained molecules.
An adiabatic process can also occur in uncontained systems like open airstreams, and the evaporation of water is one such example. (For more information about adiabatic cooling, particularly in evaporative coolers, check out this article.)

Evaporation of water is a process where heat is absorbed into water molecules. They evaporate from liquid water and become entrained in the air as a vapor, displacing some of the nitrogen and oxygen in the air. When that heat is absorbed from the air into the water, it results in lower sensible temperature, but the water is still CONTAINED IN THE AIR. This means that while the air may be cooler, it still has all the heat contained in it in the form of water vapor.
Now for the real shock:
Water vapor is NOT denser than dry air at the same temperature; it is actually less dense/lighter than dry air. If you add up the atomic masses of water (two hydrogen atoms and one oxygen) and compare that sum to an oxygen or nitrogen molecule in our atmosphere (two oxygen atoms or two nitrogen atoms), you'll find that the water molecule is lighter.

However, water vapor DOES contain more heat (enthalpy—for you nerds like me). That means that when you run the blower after a cooling cycle, the moisture on the coil and in the pan are evaporated back into the space, and depending on the RH of the air, it will lead to sensible cooling but latent gains. That means cooler but higher RH, which is due to the higher heat content of higher RH air at the same temperature.
Once again, depending on where you live, this may be positive or negative.
In Arizona or Colorado? Run that blower after the cooling cycle.
Florida? You may want to shut it off right after the cycle, or maybe 90 seconds at most; leaving the fan in the “on” position will likely result in a small increase of indoor RH.
—Bryan
P.S. – I also did a Facebook Live Video about this topic a while back:
I also wrote another article about the effect of adding or removing moisture from the air. It has more of a psychrometrics bend, so you can read that HERE if that might interest you. And there's also a short podcast about adiabatic cooling HERE.
Also, here are some great videos on the subject by Jim Bergmann:









Comments
Br’Y’an,
Thanks for all the info you put out here for us. As a newer tech I’m finding to few have an understanding of how or why things are supposed to be…. so again thanks! Also get Bergman on any chance you can…
Br’Y’an,
Thanks for all the info you put out here for us. As a newer tech I’m finding to few have an understanding of how or why things are supposed to be…. so again thanks! Also get Bergman on any chance you can…
To leave a comment, you need to log in.
Log In