Get Tech Tips
Subscribe to get free tech tips.
CO2 Booster Systems (Codenamed CO2 is Interesting and Weird)
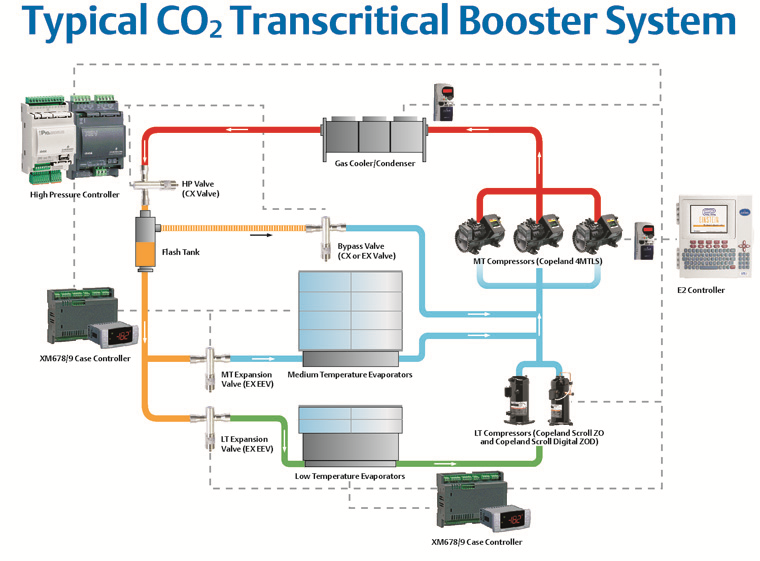
Illustration Courtesy of Emerson
CO2 is a pretty nice refrigerant.
It has zero ODP (ozone depletion potential) and a GWP (global warming potential) of 1. CO2 has been used as a refrigerant almost from the very beginning of refrigerants, and it's been making a big comeback in market refrigeration (especially in colder climates).
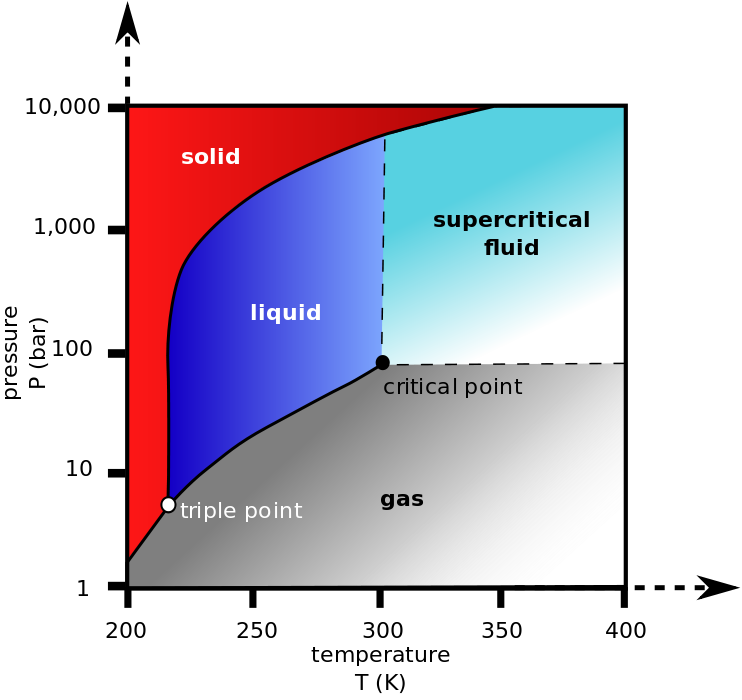
CO2 (R744) is naturally better suited for lower temperature refrigeration applications because of its low-temperature saturated state at atmospheric pressure (-109.3F). You will notice I said “saturated state” because CO2 does not “boil” at atmospheric pressure. At any pressure below 60 PSIG, CO2 goes straight from solid (dry ice) right to a vapor. That is why 60 PSIG is known as the “triple point,” or the point where CO2 could be either solid, liquid, or vapor. (For more on saturated state and what that means for technicians, check out this article here.)
Now, go to the top of the range with CO2. When you apply 1055 PSIG, the saturation temperature is 87.8°F, but go up even 1 more degree, and CO2 CANNOT be liquified. This point is known as the critical point of the substance. Whenever a substance is forced beyond its critical point, it becomes what is known as a supercritical fluid and has properties that are unique to this state, but it is certainly not a liquid. You can learn more in this Emerson article about CO2 as a refrigerant and in this Hussman CO2 transcritical system training manual.
In a transcritical (trans means beyond or through, so transcritical means “beyond critical”) booster refrigeration system, the low-temp portion of the system operates using its own compressors that “boost” the refrigerant from the low temp side and discharge into the suction of the medium temp side. The high-stage compressors then pressurize the CO2 (R744) above its critical pressure/temperature.
What is traditionally called a condenser becomes a gas cooler and decreases the temperature (rejects heat from) of the discharge without actually condensing it into liquid. The cooled supercritical fluid goes through a pressure-reducing valve, where some of it condenses into liquid, and the rest remains as gas. Liquid and gas are separated in a flash tank (receiver). Pressure in this tank is usually controlled to around 450 to 500 PSIG.
It's super critical that you understand all of this.
See what I did there?
—Bryan












Comments
Isn’t the reason they phased CO2 out because of a few catastrophic disintegrations of refrigeration equipment where windings had been hit with lightning voltage, that caused the windings to disintegrate and then disintegrate the metal housings? That is how I learned about CO2 being phased out.
Isn’t the reason they phased CO2 out because of a few catastrophic disintegrations of refrigeration equipment where windings had been hit with lightning voltage, that caused the windings to disintegrate and then disintegrate the metal housings? That is how I learned about CO2 being phased out.
CBD products have honestly changed my biography! I not under any condition expected something so natural https://cobocbd.com/collections/cbd-for-sleep to carry such equanimity and peace into my daily routine. Highlight that at the same time felt overwhelming is second so much easier to carry on, and my catnap has become deeper and more refreshing. Even muscle aches after protracted days fade faster. It feels extraordinary to completely have something light to this day potent that supports both body and mind. I can’t suspect my days without CBD anymore!
CBD products have honestly changed my biography! I not under any condition expected something so natural https://cobocbd.com/collections/cbd-for-sleep to carry such equanimity and peace into my daily routine. Highlight that at the same time felt overwhelming is second so much easier to carry on, and my catnap has become deeper and more refreshing. Even muscle aches after protracted days fade faster. It feels extraordinary to completely have something light to this day potent that supports both body and mind. I can’t suspect my days without CBD anymore!
To leave a comment, you need to log in.
Log In