Get Tech Tips
Subscribe to get free tech tips.
What does “Saturated State” mean for Techs?
As we often do in these tech tips, we will start with the common and more practical explanation of saturation and then move to the more technical and nerdy explanation later.
When we say “at saturation” or “saturated” in the HVAC/R trade, we are generally referring to the refrigerant that is in the process of changing from liquid to vapor (boiling) in the evaporator or vapor to liquid (condensing) in the condenser.
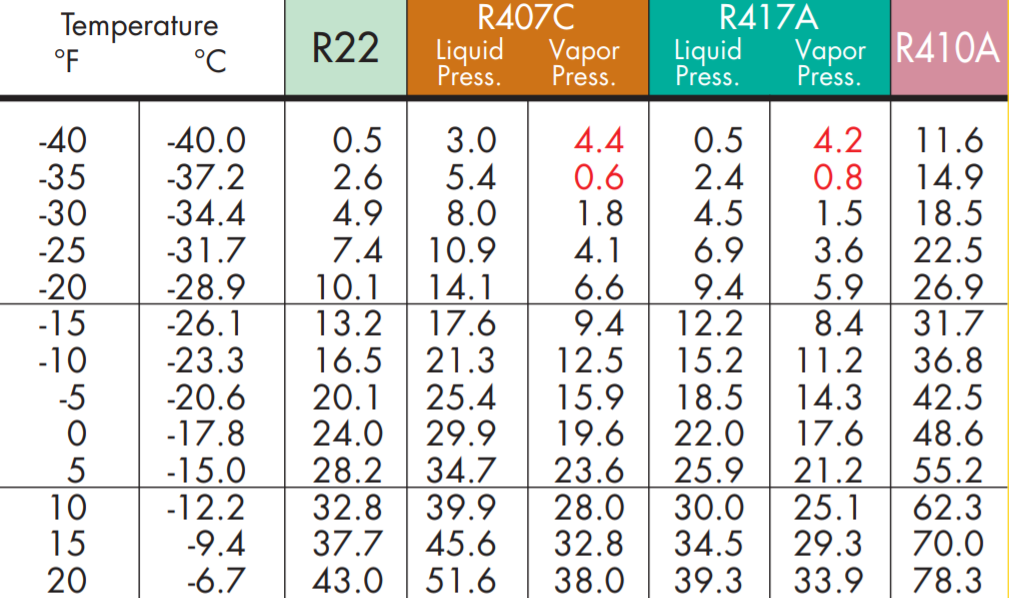
We generally look at a set of gauges or find the temperature on a PT (Pressure-Temperature) chart that matches a particular refrigerant and pressure. We call that the saturation temperature.

So, when a tech connects gauges to the liquid line (high side) of a system and looks at the needle, they will refer to the pressure in PSI and the temperature for the particular refrigerant as saturation temperature. On the gauge above, the refrigerant in the system is R22 (green scale); they would say that the pressure is 200 PSI, and the saturation temperature is 102°F.
To be even more specific, a tech might say that the condensing temperature of this system is 102°F because the saturated state is occurring during the process of condensing, in this particular case.
From a practical standpoint, when we say “saturation” in terms of a refrigeration circuit, we are referring to:
The pressure and temperature a refrigerant will be if both liquid and vapor are present at the same time and place.

One of the most common cases where we will see refrigerant at saturation is inside systems that are off and inside a refrigerant tank. If you were to connect a gauge to a tank (like this Testo 550 shown), the refrigerant pressure inside the tank would be equal to the pressure that correlates to the saturation temperature of the tank. (I know that's a mouthful, but it's really pretty simple.)
In the case of the refrigerant shown above, the room temperature is 71.9°F, and the refrigerant is R-422D. All I had to do was connect the Testo 550 and select R422D and the saturation temperature (shown above the PSI on the right), which is EXACTLY 71.9°F. In this case, we can say the saturation PRESSURE of R422D is 136.8 PSI at 71.9°F or that the saturation TEMPERATURE is 71.9°F at 136.8 PSI.
Either way, we are saying that there is both liquid and vapor present inside the tank, so it is at SATURATION—or in the saturated state if you would rather. So, as techs, we see refrigerant at saturation pressure and temperature when the system is off, inside a tank, and when it is in the midst of boiling in the evaporator or condensing in the condenser.
Now for the more in-depth explanation
I will warn you that this is a bit of a beating around bush explanation, but I'm writing the explanation I wish I had been given early on—so be patient, young grasshopper.

Let's start with a dictionary definition of saturation:
The state or process that occurs when no more of something can be absorbed, combined with, or added.
So, when something is “full” and can hold no more of something, it is said to be saturated, like a sponge saturated with water, or air saturated with water vapor, or, in this case, a liquid saturated with kinetic energy.
Many (including Wikipedia) will define saturation as the boiling point of a liquid. This definition is correct but can lead to a misunderstanding. Just because a liquid is at its boiling point doesn't mean it is actively boiling. The refrigerant in an air conditioner is technically at the boiling point when the system is completely off. The refrigerant in a tank is at saturation (so long as it has some liquid in the tank), even though the refrigerant is static (not flowing).
In nature, gasses (vapor) and liquids are free to move around and interact with one another, with the predominant pressure being atmospheric pressure (14.7 PSIA at sea level).
You may have wondered why water exposed to the air will evaporate even though it has not reached the boiling temperature. This is because the temperature of a substance is the AVERAGE kinetic energy of the molecules in a substance, not the specific kinetic energy of every single molecule. While there may not be enough energy for the entire substance to boil, there is enough energy in a few of the molecules to break free from the surface.
This is why your skin cools when sweat evaporates off of your skin. The highest energy molecules are leaving, taking themselves and their high energy ways with them!
Translation: Some molecules have more energy than others and can escape the liquid form, and we call this evaporation. This evaporation can be measured, but it happens below the boiling point. When a substance is uncontained, it results in less liquid remaining.
Translation of the Translation: If you leave water out in a pan, it will eventually disappear, even when it isn't boiling.
Now, if you put a liquid in a jar and screw the lid on, some of the molecules will escape the liquid bonds and fill the void in the jar. However, pretty soon, the jar will be at equilibrium (static) pressure with an equal number of molecules condensing back into the liquid as those that are escaping. The more active the molecules in the jar, the more pressure there will be in the jar. Since the definition of temperature is the average kinetic energy (energy of motion) of the molecules, you can translate that as, “The hotter the jar, the higher the pressure,” or “The higher the pressure, the hotter the jar.”

Different liquids have a more or less tendency to escape the liquid form (evaporate). Liquids with a very high tendency to escape will evaporate more quickly and have a higher “vapor pressure.” They are also said to be more “volatile.” Alcohol or gasoline are liquids that are more volatile and have a higher vapor pressure at atmospheric pressure than water. Hence, they disappear quickly even when the ambient temperature is below their boiling point.
Some liquids (like vacuum pump oil, for example) have a very low tendency to evaporate and have very low volatility and low vapor pressure.
Liquids with low boiling temperatures (like most refrigerants) are very volatile and have a higher vapor pressure than liquids that remain liquid at atmospheric pressure. We know that refrigerant does more than evaporate at atmospheric pressure, and at normal atmospheric temperature, it literally BOILS.
A liquid boils when the vapor pressure of the liquid matches the atmospheric pressure. At that point, the liquid molecules begin to break free rapidly, and if they are uncontained, they will simply fly away like water vapor out of an open pot.
If the molecules are boiling and contained, they will begin increasing the pressure as they boil until the temperature of the liquid no longer increases. It hits equilibrium between the vapor pressure of the liquid and the pressure inside the vessel (tank, pressure cooker, etc.).
Once the vessel can reach a state of perfect equilibrium, it may no longer be boiling, but it can still be at the boiling point. That exact POINT of equilibrium between vapor pressure and temperature is the SATURATION POINT.
So long as the pressure remains constant on a boiling or static vapor/liquid mixed substance, we can say that it is at saturation temperature because it remains at the same temperature until either:
- The pressure changes
- The substance is fully boiled
But it is important to remember that it is the vapor pressure of a liquid substance being equal to the pressure around it that results in saturation and then boiling or, in the opposite direction, condensing.
Also, evaporators should be called boilerators, but I'm doing being nerdy for now.
—Bryan
P.S. – Here's another more recent article about saturation and the pressure-temperature relationship if you're interested in approaching the topic from that angle.












Comments
“You may have wondered why water exposed to the air will evaporate even though it has not reached the boiling point? This is because the temperature of a substance is the AVERAGE kinetic energy of the molecules in a substance not the specific kinetic energy of every single molecule. While there may not be enough energy for the entire substance to boil, there is enough energy in a few of the molecules to break free from the surface.”
This is also how evaporative cooling works. Since it is the higher energy molecules that break the surface and become free, this lowers the average energy level of the remaining liquid resulting in a temperature drop. Once the temperature of the liquid drops, it becomes lower than it’s surroundings and pulls heat from it, cooling off whatever is in contact with it. Hence why sweating is a good form of heat removal and why substances with higher vapor pressures such as alcohol produce such a high cooling effect through rapid evaporation when on the skin.
Ok now I am getting really geeky.
“You may have wondered why water exposed to the air will evaporate even though it has not reached the boiling point? This is because the temperature of a substance is the AVERAGE kinetic energy of the molecules in a substance not the specific kinetic energy of every single molecule. While there may not be enough energy for the entire substance to boil, there is enough energy in a few of the molecules to break free from the surface.”
This is also how evaporative cooling works. Since it is the higher energy molecules that break the surface and become free, this lowers the average energy level of the remaining liquid resulting in a temperature drop. Once the temperature of the liquid drops, it becomes lower than it’s surroundings and pulls heat from it, cooling off whatever is in contact with it. Hence why sweating is a good form of heat removal and why substances with higher vapor pressures such as alcohol produce such a high cooling effect through rapid evaporation when on the skin.
Ok now I am getting really geeky.
To leave a comment, you need to log in.
Log In