Get Tech Tips
Subscribe to free tech tips.
Not All CFM Are Created Equal
We’re in the business of moving heat, but we have to move air to accomplish that. We use CFM (cubic feet per minute) to quantify the air we move over the indoor coil, and it helps to imagine hundreds of 1’x1’x1’ boxes of air moving over that coil each minute. (After all, 400 CFM is a common rule of thumb.)
However, CFM is only a volume measurement. It refers to the air molecules occupying a cube that is 1’x1’x1’, not the total density or weight of the molecules in that cube. While we might have a CFM target that we’re trying to hit, it’s unlikely that the air we’re moving meets the same conditions of the air for which those targets were designed. To deliver the same mass of air that those targets were designed to deliver, we need to be mindful of how that air is packed into those 1-cubic-foot boxes, not just focus on moving 400 boxes per minute and call it a day.

One cubic foot of air in Miami Beach will weigh significantly less than one cubic foot of air in Anchorage. You’re moving different concentrations of air molecules, and the blower inside the HVAC equipment will interact with these boxes of air differently. Let’s see what makes these same 1’x1’x1’ boxes of air so different from each other.
Air Has Weight & Takes Up Space
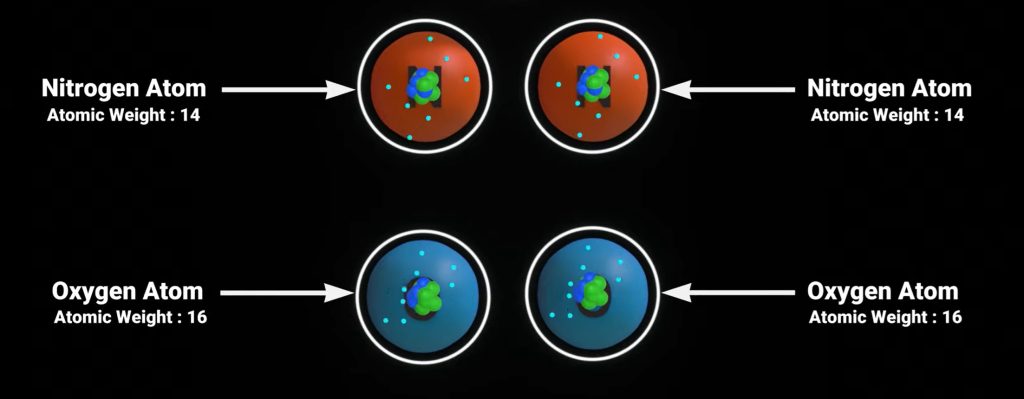
While we can’t “feel” the air suspended around us like a fleece jacket weighing down on us, it still has weight. Air is made up of “stuff.” That “stuff” is a mix of about 78% nitrogen (N2), 21% oxygen (O2), and 1% other trace elements like CO2, argon, and more.
Each element has an atomic mass, meaning it has weight (even if it’s far too small for us to feel). Each element has a nucleus, and the protons and neutrons in that nucleus have significant weight. For example, nitrogen has a weight of 14 atomic mass units or AMU (7 protons and 7 neutrons), and oxygen has a weight of 16 AMU (8 protons and 8 neutrons).
However, the weight of the molecules buzzing around us isn’t the only factor affecting how much air weighs.
Temperature
Molecules in a vapor fly past each other really quickly (as opposed to in liquids or solids). Since air is a vapor, those molecules are all moving at different speeds; moving faster indicates more heat, and moving slower indicates less heat. The average speed of those molecules is the temperature.
Hotter “boxes” of air have a higher temperature, which means that the molecules are zooming around more quickly. When these molecules move more quickly, they become less dense in that 1’x1’x1’ box. The lower density means that the box of air will have a lower weight.
Colder “boxes” of air have a lower temperature, and the molecules are a bit more sluggish. Since they’re slower and stay more closely packed, they have a higher density within those uniform boxes.
Humidity
I’m about to blow your minds. Humid air is lighter than dry air.

Remember those atomic weights?
In our atmosphere, nitrogen and oxygen exist in the diatomic form (two atoms joined together). Therefore, a nitrogen molecule has two 14-AMU nitrogen atoms for a total atomic weight of 28 AMU. Diatomic oxygen has an atomic weight of 32 AMU (16+16).
Water molecules consist of two hydrogen atoms and one oxygen atom. While oxygen molecules have an AMU of 16, hydrogen weighs only 1 AMU.
16+1+1=18

That’s a lot lighter than 28 AMU! Therefore, air with higher water vapor content (i.e., higher humidity) has less weight than dry air. That sticky feeling of humid air definitely feels “heavier” on your skin, but it’s actually lighter than air you’d find in a desert at the same temperature and altitude.
Elevation
Have you ever heard someone say that the air feels thinner at higher altitudes? Well, they’re right!
The air pressure is a lot lower at the top of a mountain than in a low-lying valley. When you’re 5,000 feet above sea level and comparing the pressure to someone who’s at sea level, the air has 5,000 more feet of space to take up between you and the person at sea level, and it’s exerting pressure in that space.
With lower air pressures, the molecules spread out a bit more, which lowers the density.
Conclusion and a Quick Analogy for Mass Flow Rate vs. Volume Flow Rate
The way we think about CFM as moving “boxes” of air is accurate and great for helping people understand what volume flow rate is, but it doesn’t tell the whole story.
Let’s say we’re in a storage unit, sliding boxes of books from one end of the unit to another. All the boxes are the same size, but one person is sliding boxes with 40 books, and you’re sliding boxes containing 39 books. You and the other person could move 10 boxes each, but they will have moved 400 books, while you will have only moved 390 books.
Sure, you moved the same number of boxes each, but you didn’t deliver the same number of books from one end of the unit to the other. The books represent the mass flow rate, and the boxes represent the volume flow rate. We’re supposed to be delivering books, not just boxes of books.
Flow rates are the same; we have to remember that we’re moving air, not just boxes of it, so not all CFM are created equal. The actual cubic feet per minute (ACFM) targets attempt to add a little more nuance to our CFM targets (often expressed in standard cubic feet per minute or SCFM, which doesn’t accurately represent field conditions most of the time). However, ACFM vs. SCFM is a topic of its own. (If you’re curious, watch this 3D animation we made; it also covers everything in this tech tip.)









Comments
To leave a comment, you need to log in.
Log In