Get Tech Tips
Subscribe to free tech tips.
Ideal Gas Rulebreakers

Breaking the Sound Barrier
So, what do you think of when you hear an “ideal gas?” R22, R12 maybe… natural? Take a look at the F-18 above. It's breaking the sound barrier, and that cloud is a shockwave. It has nothing to do with this article, but I think it's pretty darn cool!
An ideal gas is a gas that obeys the ideal gas law; it's ideal because it's good at following rules. These ideal gases walk in a straight line, they don't run on the playground, and they never fish without a proper permit. In all seriousness, an ideal gas behaves predictably with changes in volume, pressure, temperature, and mass.
The problem is that a truly “ideal” gas really doesn't exist.
While many gases behave close to ideal at normal temperatures, there is no gas that obeys the ideal gas laws in all conditions.
The ideal gas law is:
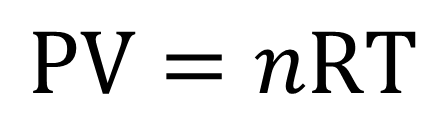
P= Absolute Pressure (gauge pressure + atmospheric pressure)
V = Volume (How much space the gas occupies)
n = Mass measured in “moles” (the number of molecules)
R = The universal gas constant (varies depending on the units of measure being used Example: [lbf ft/(lb mol oR)]= 8.3145 )
T = Absolute Temperature (temperature in a scale that starts at absolute zero like Kelvin or Rankine)
The ideal gas law is really a combination of several different laws into one.
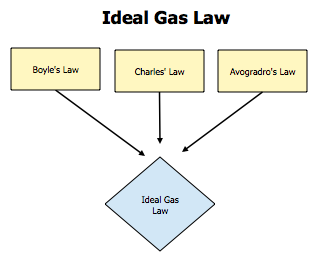
The result is that many gases in our work behave in about the same way with changes in mass, volume, temperature, and pressure. That's the case because the primary force at play in a nearly ideal gas, like nitrogen or CO2, is simply the velocity of the molecules bouncing around in the container and against one another like tiny little ping pong balls.
If the molecules react or interact with one another through attraction or repulsion due to their intermolecular forces, they can cease to behave as an ideal gas. A perfect example is when a gas is in contact with its liquid form (saturation), it no longer obeys the gas laws. That is why most gases behave more and more like an ideal gas the hotter they get (within a range). The hotter they are, the greater the force of molecular velocity (temperature) will be relative to the intermolecular interaction of the molecules.
Once the gas gets to the “supercritical” state, all bets are off once again. So, like most good kids, even the most ideal gases have their limits. If pushed, they become little molecular rebels.
—Bryan









Comments
To leave a comment, you need to log in.
Log In