Get Tech Tips
Subscribe to get free tech tips.
A Common-Sense Look at Temperature Conversions

Before we convert temperature scales, let's take a step back and think about what temperature is in the first place.
Temperature is proportional to the average kinetic energy of the random microscopic motions of the constituent microscopic particles, such as electrons, atoms, and molecules.
Translation: Temperature is the average “movement energy” of the molecules in a substance
Higher temperature means there is more average heat ENERGY when compared to the same mass of the same substance at a lower temperature. While there is no limit to how high the temperatures of matter can go (at least that science is aware of), there is a bottom limit, and that is the point at which there is NO HEAT, and therefore no molecular or atomic motion. That point of no energy is called ABSOLUTE ZERO. (To learn more about absolute zero and its role in thermodynamics, please check out our article on the third law of thermodynamics.)
Absolute zero is a theoretical point because it has never been (and likely will never be) achieved. For most of us, absolute zero has no real application, and this is why our most common temperature scales are tied to freezing and boiling instead of absolute zero.
Anyone can make up their own temperature scale. All you need to do is pick a “zero” point based on a known value (say, boiling or freezing water at atmospheric pressure) and then decide on the size of a degree.
In the “Fahrenheit” scale, a guy named Daniel Gabriel Fahrenheit decided to make a temperature scale. The coldest “constant” he had at his disposal was a water and brine solution, so he called that 0°F. He then used the body temperature of an “average healthy man” and somewhat arbitrarily called that 96°F. This dictated the “size” of the Fahrenheit degree as well as the zero point. From there, he ascertained that on his scale, water froze at 32°F and boiled at 212°F at sea level (14.7 PSIA).
The Celsius scale (often called Centigrade by old-timers) logically used the freezing and boiling of water as the 0° and 100° points. This established a logical starting point at the freezing of water and a LARGER degree size than the Fahrenheit scale.
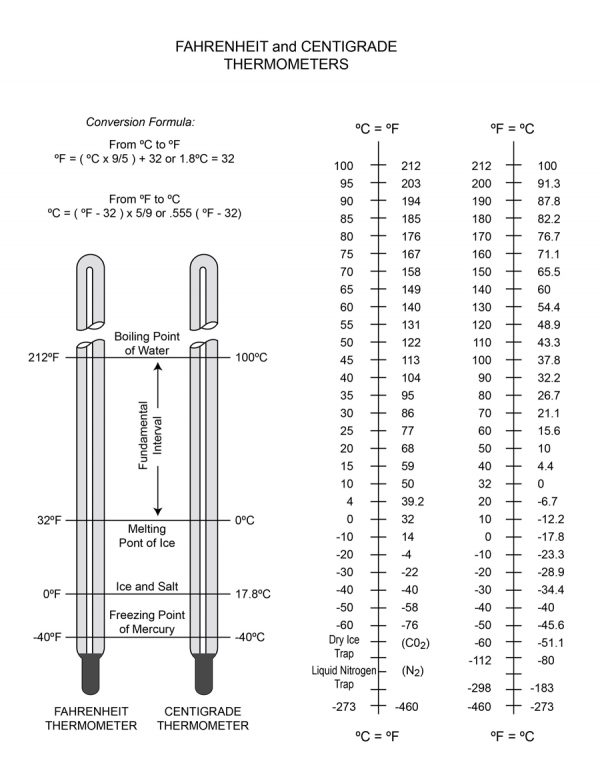
This means that when you are converting a temperature—say, 75°F to Celsius—you first subtract 32° to normalize for the different starting points of the scale, and THEN you multiply by .555 (I prefer decimals because fractions don't work on most calculators), so 75°F = 23.865°C.
But be careful…
When converting a temperature comparison or differential, you must skip the +/- 32° part of the process. In those cases, you are only converting the SIZE of the degree, not its location on the scale!
That means that if we are discussing 10°F of subcooling, we would simply multiply it by .555 to see that it is 5.55°C of subcooling.
HOWEVER…
Scientists didn't like the old Fahrenheit and Celsius systems because they are scientifically nonsensical. “There is NO SUCH THING as negative heat!” exclaimed the angry Mr. Kelvin & Mr. Rankine (at least, that's how they do in my imagination). So, they invented scales where zero is ABSOLUTE ZERO so that there are no negative numbers. The Kelvin scale starts with 0 at absolute zero and uses the Celsius degree size, and Rankine starts at absolute zero and uses the Fahrenheit degree size.
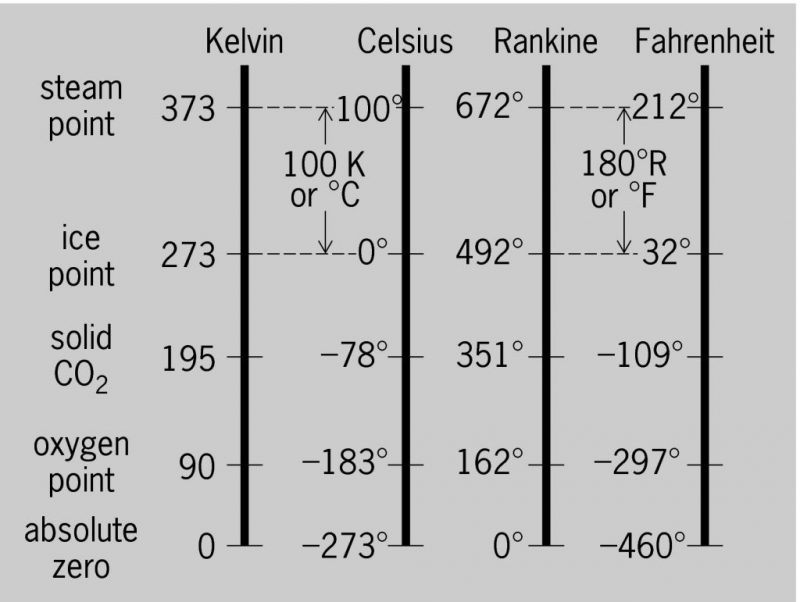
So, whenever we make a conversion from Fahrenheit to Celsius on the SCALE, we show the converted temperature as °C. However, when making a conversion that is simply a comparison or a differential (DTD, delta T, CTOA, superheat, subcool, etc.), we show it as kelvins (K) to help us differentiate.
Confusing, huh?
—Bryan












Comments
Hey Bryan,
your article are really informative and Insightful
Thank You.
Br,
Ammer
Owner – HitechDuct
Hey Bryan,
your article are really informative and Insightful
Thank You.
Br,
Ammer
Owner – HitechDuct
Did you know your body is wired to reciprocate to CBD? Every magnanimous has an endocannabinoid pattern (ECS) that influences atmosphere, have a zizz, and unsusceptible response. CBD is unequalled because it doesn’t bind immediately to receptors like THC does; in preference to, it encourages the body to utilization its own endocannabinoids more effectively. It’s like a tune-up in the interest your biological signaling, promoting efficiency and wealth
Did you know your body is wired to reciprocate to CBD? Every magnanimous has an endocannabinoid pattern (ECS) that influences atmosphere, have a zizz, and unsusceptible response. CBD is unequalled because it doesn’t bind immediately to receptors like THC does; in preference to, it encourages the body to utilization its own endocannabinoids more effectively. It’s like a tune-up in the interest your biological signaling, promoting efficiency and wealth
I’ve been using https://www.nothingbuthemp.net/products/lions-mane-mushroom-gummies-for-focus constantly in regard to on the other side of a month nowadays, and I’m indeed impressed during the uncontested effects. They’ve helped me feel calmer, more balanced, and less solicitous everywhere the day. My forty winks is deeper, I wake up refreshed, and straight my core has improved. The trait is excellent, and I worth the common ingredients. I’ll obviously preserve buying and recommending them to person I know!
I’ve been using https://www.nothingbuthemp.net/products/lions-mane-mushroom-gummies-for-focus constantly in regard to on the other side of a month nowadays, and I’m indeed impressed during the uncontested effects. They’ve helped me feel calmer, more balanced, and less solicitous everywhere the day. My forty winks is deeper, I wake up refreshed, and straight my core has improved. The trait is excellent, and I worth the common ingredients. I’ll obviously preserve buying and recommending them to person I know!
Bryan,
I’m looking at the technical spec sheet for an Emerson / Copeland scroll compressor. The Superheat and Subcool values are listed as 11.1(K) and 8.3(K). So, in this case these values are actually degrees F and the (K) indicates a differential or comparison?
Bryan,
I’m looking at the technical spec sheet for an Emerson / Copeland scroll compressor. The Superheat and Subcool values are listed as 11.1(K) and 8.3(K). So, in this case these values are actually degrees F and the (K) indicates a differential or comparison?
From your article it is clear to see that the infamous 5/9 or 9/5 fractions comes from the fact the there are 100 Centigrade degrees (Centigrade means a hundred parts) to every 180 Fahrenheit degree and 180 Fahrenheit degrees to every Centigrade degree. And the reason for adding or subtracting 32 is for comparison of values (so both start at zero). I never noticed that before. It will make the conversion much easier for me to remember in the future. Now all I have to do is remember that the value I’m looking for (e.g., 100 for Centigrade) goes on top of the fraction and that the scale shifts up (+) 32 to get Fahrenheit and down (-) 32 to get Centigrade. Thanks.
From your article it is clear to see that the infamous 5/9 or 9/5 fractions comes from the fact the there are 100 Centigrade degrees (Centigrade means a hundred parts) to every 180 Fahrenheit degree and 180 Fahrenheit degrees to every Centigrade degree. And the reason for adding or subtracting 32 is for comparison of values (so both start at zero). I never noticed that before. It will make the conversion much easier for me to remember in the future. Now all I have to do is remember that the value I’m looking for (e.g., 100 for Centigrade) goes on top of the fraction and that the scale shifts up (+) 32 to get Fahrenheit and down (-) 32 to get Centigrade. Thanks.
To leave a comment, you need to log in.
Log In