Get Tech Tips
Subscribe to free tech tips.
Air Flow and Ice – A Thought Experiment
Imagine a glass of ice sitting on a table.
Now, imagine you place a lid on the glass so that all the water and ice are contained in the glass.
If the ice and water are well mixed, the water and ice will both be at 32°F because the ice is slowly changing state from ice to water, which we call melting. Because this is happening at atmospheric pressure, we can know what temperature this will occur at. The heat being transferred is going toward melting the ice rather than changing the water temperature. We call that latent heat. (For more information on the basics of heat transfer, check out this guide.)

Let's say the temperature in the room is 75°F. In this scenario, heat leaves the air molecules as they contact the exterior of the glass, and heat moves through the glass into the water and ice. That happens pretty slowly because glass is a pretty good insulator, but this heat still moves from hotter to colder.
This movement of heat from the air to the glass exterior occurs THROUGH the glass via conduction. To learn more about conduction, convection, and radiation, check out this video.
What happens if we blow air toward the glass? What changes?
If we move more air over the side of the glass, we deliver more air molecules to the glass via convection, but it doesn't change the fact that the heat makes it through the walls and into the glass via conduction.
By delivering more air to the glass, we warm the outside of the glass more, which causes the water to melt inside the glass faster. In other words, more air over the glass means more heat transfer even though we didn't change the temperature of the water or the air.
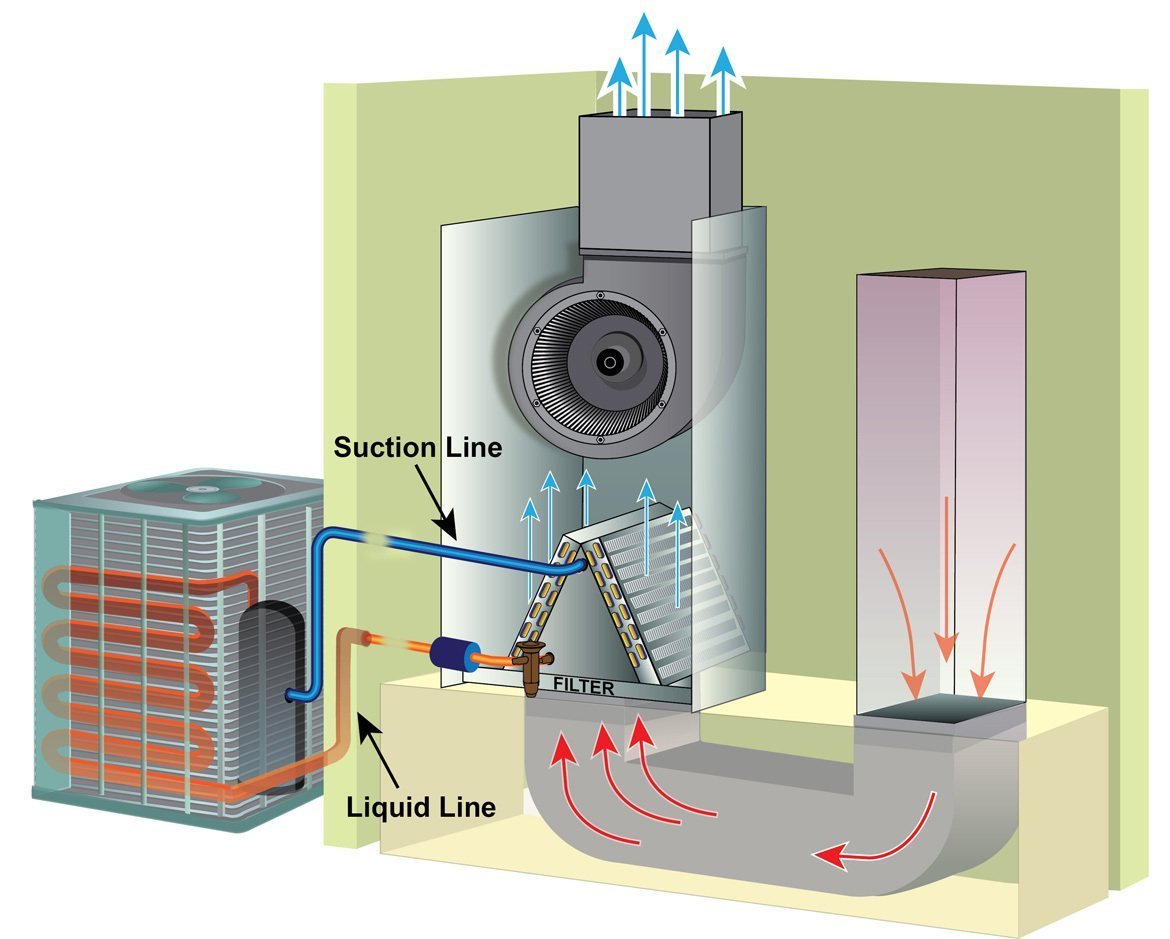
This same basic thing happens inside an evaporator and condenser coil; when we increase the flow of air, we also increase the transfer of heat through the walls of the copper tubing in the coils. In the condenser, more airflow increases the heat rejection out of the refrigerant, and more heat is gained in the evaporator.
Because the refrigerant circuit is dynamic (refrigerant moving) and under pressure, more or less heat entering or leaving the system impacts the process and changes the pressures of the refrigerant inside.
If we move less air over the condenser, the pressure on the high side increases. If we reduce the air over the evaporator coil, less heat enters the circuit, and pressures drop.
This is a basic picture for you to consider the next time you see high or low system pressures and how the coil airflow impacts heat transfer.
A more advanced but similar thought experiment considers what would happen if the evaporator coil had no fins.
—Bryan










Comments
To leave a comment, you need to log in.
Log In